Jessica MacDonald

Jessica MacDonald
Associate Professor
CONTACT
Biology
360 Life Sciences Complex
Email: jemacdon@syr.edu
Office: 315.443.5837
PROGRAM AFFILIATIONS
Biochemistry
Biotechnology
Degrees
- Postdoctoral Fellow, Department of Stem Cell and Regenerative Biology, Harvard University (2008-2015)
- Ph.D., Neuroscience, University of British Columbia (2008)
- Hons. B.Sc., University of Toronto (2001)
Social/Academic Links
Genetic and epigenetic mechanisms regulating neuronal development and function, with a particular interest in disruptions in these mechanisms that cause neurodevelopmental and cognitive disorders.
Research Spotlight
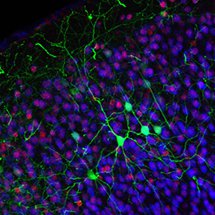
Research in the MacDonald lab focuses on understanding how transcriptional and epigenetic regulators act at the interface between genes and the environment, translating exposure to extrinsic factors into distinct neuronal phenotypes. Extensive research into neuronal disorders highlights that genetics alone fail to account for many of these complex syndromes, leading to growing evidence that epigenetics and gene-environment interactions play critical roles in neuronal development and cognitive function. We employ in vivo and in vitro animal models, and an array of molecular and cellular biological and microscopy approaches, to study how different genetic disruptions and environmental/ nutritional factors modify neuronal development and function, throughout the lifespan of an organism.
One current area of research focuses on the neurological disorder Rett Syndrome, caused by mutations in the epigenetic regulator MeCP2. Focusing on neurons within the neocortex, we are identifying molecular and cellular pathways that are disrupted in this disorder, and investigating potential therapeutic interventions of these perturbations, including by defined dietary supplementation. The ultimate goal of this research is to understand how life-long environmental exposures intersect with fixed genetic susceptibilities to alter neuronal development and cognitive function.
- Wagner, N.W.+, Siththanandan, V.B.+, Sinha, A.+, Kowalchuk, A.M., MacDonald, J.L.* and Tharin, S*. (2022) miR-409-3p represses Cited2 to refine neocortical layer V projection neuron identity. Frontiers in Neuroscience. 16:931333 +Co-first authors; *Co-senior and corresponding authors. DOI:10.3389/fnins.2022.931333/full
- Ribeiro, M.C. and MacDonald, J.L. (2022) Vitamin D modulates cortical transcriptome and behavioral phenotypes in an Mecp2 heterozygous Rett syndrome mouse model. Neurobiology of Disease.165:105636. DOI: 10.1016/j.nbd.2022.105636
- MacDonald, J.L.*, Tharin, S., Hall, S.E. (2022) Epigenetic regulation of nervous system development and function. Neurochemistry International. 152:105249 (Invited Editorial). DOI: https://doi.org/10.1016/j.neuint.2021.105249
- Marballi, K.K. and MacDonald, J.L.* (2021) Proteomic and transcriptional changes associated with MeCP2dysfunction reveal nodes for therapeutic intervention in Rett syndrome. Neurochemistry International.148:105076 (Review Article with novel analyses of published data sets). DOI: https://doi.org/10.1016/j.neuint.2021.105076
- Wagner, N.R. and MacDonald, J.L.* (2021) Atypical neocortical development in the Cited2 conditional knockout leads to behavioral deficits associated with neurodevelopmental disorders. Neuroscience.455:65-71 https://doi.org/10.1016/j.neuroscience.2020.12.009
- Ribeiro, M.C., Moore, S.M., Kishi, N., Macklis, J.D.*, and MacDonald, J.L.* (2020) Vitamin D supplementation rescues aberrant NF-kappaB pathway activation and partially ameliorates Rett syndrome phenotypes in Mecp2 mutant mice. eNeuro. 7(3):1-18. DOI: 10.1523/ENEURO.0167-20.2020
- Ribeiro, M.C. and MacDonald, J.L.* Sex Differences in Mecp2-mutant Rett syndrome model mice. (2020) Brain Research. 1729:146644. DOI: 10.1016/j.brainres.2019.146644
Complete list of publications: https://www.ncbi.nlm.nih.gov/sites/myncbi/jessica.macdonald.1/bibliography/52634951/public/?sort=date&direction=ascending
