Cutting Edge
Talking milk cartons and superconducting batteries? They may be closer than you think
Imagine knowing if food was spoiled just by looking at the packaging, or having a phone battery that never overheats and lasts much longer between charges. These possibilities could eventually be realized, thanks to ongoing research in the Department of Chemistry.
Davoud Mozhdehi, assistant professor of chemistry, is working on an autonomous synthetic material that could create what he calls “smart plastics.” Steluța A. Dincă, research assistant professor of chemistry, and a team of chemists are working to develop a superconductor that could store vast amounts of energy and make the electrical power grid much more efficient.
Teaching plastics to “speak”
According to notreallyexpired.com, 40% of the food produced in the United States goes uneaten each year, resulting in 160 billion pounds of wasted food. At the same time, 37 million Americans struggle with hunger, according to feedingamerica.org. A new kind of plastic could be part of the solution to the problem of food waste.
Much of that waste is due to confusion over the sell-by and use-by date labels and whether or not food is actually expired. Imagine having a milk container programmed to analyze its contents and alert the consumer that the milk is spoiled. Such a product could eliminate the need for the smell test and our uncertainties about the use-by date stamped on packaging and ensure that the milk we are consuming is safe to drink.

Most food is thrown away in America not because it is rotten, like these berries, but because of confusion over sell-by dates.
To make this vision a reality, researchers in Assistant Professor of Chemistry Davoud Mozhdehi’s lab are working to develop a new form of synthetic polymer that can consume fuel (i.e., the potentially spoiled food), change after eating and emit a visual signal. Similar to the way the human body produces heat after eating food, these new synthetic polymers, known as Thermally Energized Self-Regulating Materials System (ThERMoS), will generate heat after consuming energy. The heat generated would then create a visual cue for the consumer.
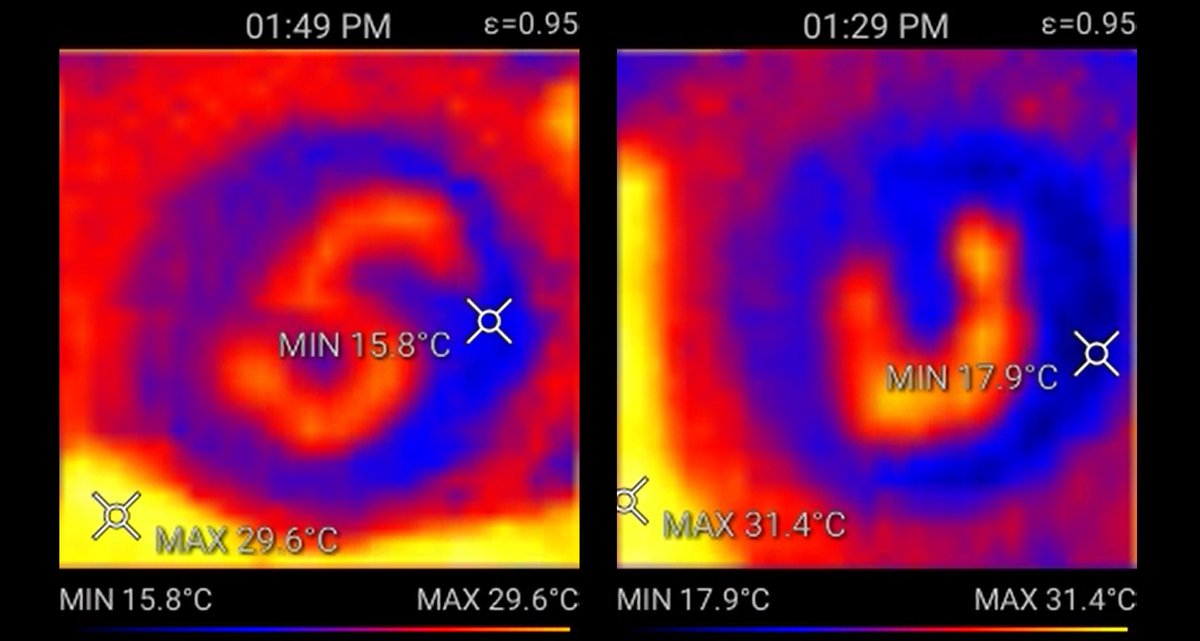
The reddish-orange "S" and "U," for Syracuse University, indicate where the polymer emitted heat after consuming chemical fuel.
Mozhdehi captured photographic proof of the process on a petri dish with an infrared camera. The image shows a polymer emitting heat after consuming chemical fuel, shown by a reddish-orange color.
A seismic shift
Plastics were created to take the place of metal and glass, both static materials. Mozhdehi and his team are essentially training plastics to act in a much different manner—interacting with their environment and converting chemical energy into usable feedback.
“Our goal is to make plastics that make a decision by using the energy acquired from eating,” says Mozhdehi. “This is a profound transformation in the use and properties of plastics.”
In the case of the spoiled milk, when bacteria begin to grow, the trained plastic would change color to alert the consumer that it is no longer safe to drink. In time, the response generated from this kind of smart plastic interaction could make use-by and sell-by dates much more accurate, reassuring consumers that there is no need to discard that unopened package, and no need to add to the food waste crisis.
Although it may be years until this technology is viable on the consumer level, thanks to Mozhdehi’s research and others in BioInspired Syracuse: Institute for Material and Living Systems, what sounds like science fiction may someday be a reality.
Mozhdehi’s research is being funded through a grant from the American Chemical Society’s Petroleum Research Fund (PRF). PRF grants are awarded to fundamental research in the petroleum field. Since most plastics are derived from petroleum, research that could advance polymer technology is supported by the PRF.
Resistance is futile
Whenever you use an electrical device, you encounter electrical resistance. This resistance causes your computer to run hot, batteries to heat up and degrade, and electrical motors in appliances to overheat.
What causes this resistance? The electrons flowing through the copper wires in your house collide with the wires’ copper atoms, like a tiny, quantum mechanical game of billiards. In each collision, the electrons lose some of their energy. These collisions transfer energy from the moving electrons to the copper atoms, causing resistance to the overall flow of electrons in the form of friction. This friction is the reason why wires become warm to the touch.
It’s also why energy costs what it does. According to the U.S. Energy Information Administration, an average of 5% of the energy generated by power plants is lost due to electrical resistance just in the power lines.

The holy grail of power
Can a better material be created that can both conduct electricity without resistance - a room-temperature superconductor – and be used beyond just the experimental laboratory?
It’s been the holy grail of researchers worldwide for decades. Today, a team of researchers in A&S is working on a superconducting material that just might overcome the stumbling blocks around this idea.
One of the enduring challenges in creating economical room-temperature superconductive materials is their lack of practicality outside the lab. For example, only certain metals, ceramics and organic salts are known to conduct electricity with no resistance, and they must be cooled to extremely low temperatures or incredibly high pressures before their resistance drops to zero.
For example, some cuprates—ceramic materials made of copper and oxygen—become superconductors at around 140 K (-140 C, -208 F). That’s easy to achieve in a laboratory, but not something to design a power grid around. Given the cost of the equipment needed to reach superconductivity in known materials, researchers around the world keep searching for the perfect room-temperature conductor.
A possible solution
The organic polymer polyacetylene could be one solution. The interdisciplinary A&S team—which includes a physicist, Research Assistant Professor Steluța A. Dincă, and chemists Professor Emeritus Bruce S. Hudson, Professor Michael B. Sponsler, Research Assistant Professor Damian G. Allis and graduate student Michael D. Moskowitz—is developing a method for producing polyacetylene crystals, which may exhibit metal-like conductivity and, perhaps, room-temperature superconductivity.

March 2020 issue of Chemistry of Materials
The group has developed a method known as elimination-condensation inclusion polymerization to produce aligned polyacetylene chains. If the team succeeds in creating long polyacetylene wires, the wires might have the potential to carry record quantities of electrical current for their type and size.
The team’s research article, “Preparation of Ordered Polyacetylene by Solid-State Polymerization in Nanoscale Confinement,” was featured on the cover of the March 2020 issue of the scientific journal Chemistry of Materials. Computations using quantum chemical methods were performed on the University’s OrangeGrid computer network.
Dincă says that she and her colleagues will look to study the electrical properties of the polyacetylene chains to determine how conductive—or potentially superconductive—this material might be.
Better batteries
In addition to being an important step in unraveling the mystery of room-temperature superconductivity, polyacetylene could be used as the base material to make energy storage devices, like those used in houses that are powered by wind and solar energy, and rechargeable batteries, like the ones used in electric vehicles.
A battery made from polyacetylene wires would take the shape of a superconducting ring. The way that energy is stored in such a device is quite different from that of a common lithium-ion battery. In the case of lithium-ion, the electrical energy is stored by way of a chemical potential. The use, or discharge, of the battery involves lithium ions moving across a membrane, while the recharging of the battery occurs as the lithium ions are forced back across the membrane when, for instance, you plug the charger into an outlet.
Battery always included
In a superconducting battery, an electrical current is driven into the ring of material and, without resistance, this current can flow in this ring indefinitely without loss. The electrical energy obtained for powering devices comes from the magnetic field generated by the flow of current in the ring, and the recharge cycle is incredibly short, compared to the same process in a chemical battery.

As to how this material can improve battery life, Sponsler says, “As long as the material in the battery remains superconductive, the life of the battery would be infinite. In other words, the energy would remain there ready to be tapped whenever. Once the energy is tapped, the energy in the battery would be reduced by however much is used, but it could be recharged to replace this energy.” The exact amount of energy that could be stored in one of these polyacetylene magneto-batteries is still hard to estimate without further study.
“It would be amazing,” Dincă says. “If this material is a room temperature superconductor, we will look into developing a magnetic energy storage device (SMES) based on this material. It will not require a cooling system, will be smaller size and lighter, and hence will have a lower overall cost than current chemical batteries.”
From a superconducting wire that could carry 100 times more current than a same-sized copper wire, to a device that could rapidly absorb or deliver high quantities of energy, the work being done in A&S could play a crucial role in improving electric power and energy storage industries of the future.

